Keq, Kw and pH
As H2O is the medium of biological systems one must consider the
role of this molecule in the dissociation of ions from biological molecules.
Water is essentially a neutral molecule but will ionize to a small degree. This
can be described by a simple equilibrium equation:
H2O <-------> H+ + OH- Eqn. 1
This equilibrium can be calculated as for any reaction:
Keq = [H+][OH-]/[H2O]
Eqn. 2
Since the concentration of H2O is very high (55.5M) relative to
that of the [H+] and [OH-], consideration of it is
generally removed from the equation by multiplying both sides by 55.5 yielding a
new term, Kw:
Kw = [H+][OH-] Eqn.
3
This term is referred to as the ion product. In pure water, to which no
acids or bases have been added:
Kw = 1 x 10-14 M2 Eqn. 4
As Kw is constant, if one considers the case of pure water to
which no acids or bases have been added:
[H+] = [OH-] = 1 x 10-7 M Eqn. 5
This term can be reduced to reflect the hydrogen ion concentration of any
solution. This is termed the pH, where:
pH = -log[H+] Eqn.
6back to
the top
pKa
Acids and bases can be classified as proton donors and proton acceptors,
respectively. This means that the conjugate base of a given acid will carry a
net charge that is more negative than the corresponding acid. In biologically
relavent compounds various weak acids and bases are encountered, e.g. the acidic
and basic amino acids, nucleotides, phospholipids etc.
Weak acids and bases in solution do not fully dissociate and, therefore,
there is an equilibrium between the acid and its conjugate base. This
equilibrium can be calculated and is termed the equilibrium
constant = Ka. This is also sometimes referred to as the
dissociation constant as it pertains to the dissociation of protons from acids
and bases.
In the reaction of a weak acid:
HA <-----> A- + H+ Eqn.
7
the equlibrium constant can be calculated from the following equation:
Ka = [H+][A-]/[HA] Eqn. 8
As in the case of the ion product:
pKa = -logKa Eqn.
9
Therefore, in obtaining the -log of both sides of the equation describing
the dissociation of a weak acid we arrive at the following equation:
-logKa = -log[H+][A-]/[HA] Eqn. 10
Since as indicated above -logKa = pKa and taking into
account the laws of logrithms:
pKa = -log[H+] -log[A-]/[HA] Eqn. 11
pKa = pH -log[A-]/[HA] Eqn.
12
From this equation it can be seen that the smaller the pKa value
the stronger is the acid. This is due to the fact that the stronger an acid the
more readily it will give up H+ and, therefore, the value of [HA] in
the above equation will be relatively small.
back to
the top
The Henderson-Hasselbalch Equation
By rearranging the above equation we arrive at the Henderson-Hasselbalch equation:
pH = pKa + log[A-]/[HA] Eqn.
13
It should be obvious now that the pH of a solution of any acid (for which
the equilibrium constant is known, and there are numerous tables with this
information) can be calculated knowing the concentration of the acid, HA, and
its conjugate base [A-].
At the point of the dissociation where the concentration of the conjugate
base [A-] = to that of the acid [HA]:
pH = pKa + log[1] Eqn.
14
The log of 1 = 0. Thus, at the mid-point of a titration of a weak acid:
pKa = pH Eqn. 15
In other words, the term pKa is that pH at which an equivalent
distribution of acid and conjugate base (or base and conjugate acid) exists in
solution.
back to
the top
Buffering
It should be noted that around the pKa the pH of a solution does
not change appreciably even when large amounts of acid or base are added. This
phenomenon is known as buffering. In most
biochemical studies it is important to perform experiments, that will consume
H+ or OH- equivalents, in a solution of a buffering agent
that has a pKa near the pH optimum for the experiment.
back to
the top
Clinical Significance of Blood Buffering
The pH of blood is maintained in a narrow range around 7.4. Even relatively
small changes in this value of blood pH can lead to severe metabolic
consequences. Therefore, blood buffering is
extremely important in order to maintain homeostasis. Although the blood
contains numerous cations (e.g., Na+, K+, Ca2+
and Mg2+) and anions (e.g., Cl-,
PO43- and SO42-) that can, as a
whole, play a role in buffering, the primary buffers in blood
are hemoglobin in erythrocytes and bicarbonate ion (HCO3-)
in the plasma. Buffering by hemoglobin is accomplished by ionization
of the imidazole ring of histidines in the protein.
The formation of bicarbonate ion in blood from CO2 and
H2O allows the transfer of relatively insoluble CO2 from
the tissues to the lungs, where it is expelled. The major source of
CO2 in the tissues comes from the oxidation of ingested carbon
compounds.
Carbonic acid is formed from the reaction of dissolved CO2 with
H2O. The relationship between carbonic acid and bicarbonate ion
formation is shown in equations 16 and 17.
CO2 + H2O <------>
H2CO3 Eqn. 16
H2CO3 <-------> H+ +
HCO3- Eqn. 17
The reactions shown in equations 16 and 17 occur predominately in the
erythrocytes, since nearly all of the CO2 leaving tissues via the
capillary endothelium is taken up by these cells. This reaction is catalyzed by
carbonic anhydrase. Ionization of carbonic acid then occurs
spontaneously (as shown in equation 17), yielding bicarbonate ion.
Carbonic acid is a relatively strong acid with a pKa of 3.8.
However, carbonic acid is in equilibrium with dissolved CO2.
Therefore, the equilibrium equation for the sum of equations 16 and 17 requires
a conversion factor, since CO2 is a dissolved gas. This factor has
been shown to be approximately 0.03 times the partial pressure of CO2
(PCO2). When this is entered into the Henderson-Hasselbalch
equation:
pH = 6.1 + log
[HCO3-/(0.03)(PCO2)] Eqn. 18
where the apparent pKa for bicarbonate formation, 6.1, has been introduced
into equation 18.
The PCO2 in the peripheral tissues is approximately 50mm
Hg, whereas in the blood entering the peripheral tissues it is approximately
40mm Hg. This difference results in the diffusion of CO2 from the
tissues into the blood in the capillaries of the periphery. When the
CO2 is converted to H2CO3 within the
erythrocytes and then ionizes, the hydrogen ions (H+) are buffered by
hemoglobin. The production of H+ ions within erythrocytes and their
subsequent buffering by hemoglobin results in a reduced affinity of hemoglobin
for oxygen. This leads to a release of O2 to the peripheral tissues,
a phenomenon is termed the Bohr effect.
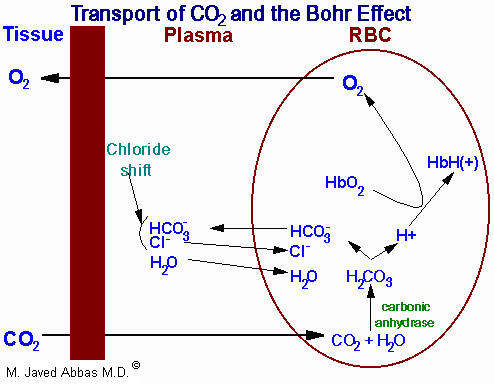 |
| Representation of the transport of
CO2 from the tissues to the blood with delivery of
O2 to the tissues. The opposite process occurs when
O2 is taken up from the alveoli of the lungs and the
CO2 is expelled. All of the processes of the transport of
CO2 and O2 are not shown such as the formation and
ionization of carbonic acid in the plasma. The latter is a major mechanism
for the transport of CO2 to the lungs, i.e. in the plasma as
HCO3-. The H+ produced in the plasma by
the ionization of carbonic acid is buffered by phosphate
(HPO42-) and by proteins. Additionally, some 15% of
the CO2 is transported from the tissues to the lungs as hemoglobin carbamate as shown in Eqn. 19.
|
As CO2 passes from the tissues to the plasma a minor amount of
carbonic acid takes form and ionizes. The H+ ions are then buffered
predominantly by proteins and phosphate ions in the plasma. As the concentration
of bicarbonate ions rises in erythrocytes, an osmotic imbalance occurs. The
imbalance is relieved as bicarbonate ion leaves the erythrocytes in exchange for
chloride ions from the plasma. This phenomenon is known as the chloride shift which is also shown in the diagram above.
Therefore, the majority of the bicarbonate ion formed as CO2 leaves
the peripheral tissues is transported by the plasma to the lungs.
Around 15% of CO2 transport from the tissues to the lungs occurs
through a reversible combination with non-ionized amino groups (-NH2)
of hemoglobin forming what is termed hemoglobin
carbamate.
Hemoglobin-NH2 + CO2 <--->
Hemoglobin-NH-COO- + H+ Eqn.
19
The formation of hemoglobin carbamate results in a reduced affinity of
hemoglobin for O2 thus favoring dissociation of bound oxygen in the
tissues where the concentration of CO2 is high. The process is
reversed when the erythrocytes enter the lungs and the partial pressure of
O2 is elevated.
The partial pressure of O2 (PO2) in the
pulmonary alveoli is higher than the PO2 of the entering
erythrocytes that contain predominantly deoxygenated hemoglobin. This increased
PO2 leads to oxygenation of hemoglobin and release of
H+ ions from the hemoglobin. The released H+ ions combine
with the bicarbonate ions to form H2CO3. Cellular
carbonic anhydrase then catalyzes the reverse of reaction 17,
leading to release of CO2 from erythrocytes. Owing to the
PCO2 gradient (described above), the CO2 diffuses
from the blood to the alveoli where it is expelled.
The great utility of bicarbonate as a physiological buffer stems from the
fact that if excess acid is added to the blood the concentration of bicarbonate
ion declines and the level of CO2 increases. The CO2 then
passes from capillaries in the pulmonary alveoli and is expelled. As a
consequence, the H+ ion concentration drives reaction 17 to the left
and bicarbonate ion acts as a buffer until all of the hydrogen ion is consumed.
Conversely, when excess base is added to the blood, CO2 is consumed
by carbonic acid and replaced by metabolic reactions within the body.
If blood is not adequately buffered, the result may be metabolic acidosis or metabolic
alkalosis. These physiological states can be reached if a metabolic
defect results in the inappropriate accumulation or loss of acidic or basic
compounds. These compounds may be ingested , or they may accumulate as metabolic
by-products such as acetoacetic acid and lactic acid. Both of these will ionize,
thereby increasing the level of H+ ions that will in turn remove
bicarbonate ions from the blood and alter blood pH. The predominant defect in
acid or base elimination arises when the excretory system of the kidneys is
impaired. Alternatively, if the lungs fail to expel accumulated CO2
adequately and CO2 accumulates in the body, the result will be
respiratory acidosis. If a decrease in
PCO2 within the lungs occurs, as during hyperventilation, the
result will be respiratory alkalosis.
back to
the top
Ampholytes, Polyampholytes, pI and Zwitterion
Many substances in nature contain both acidic and basic groups as well as
many different types of these groups in the same molecule. (e.g. proteins).
These are called ampholytes (one acidic and one
basic group) or polyampholytes (many acidic and
basic groups). Proteins contains many different amino acids some of which
contain ionizable side groups, both acidic and basic. Therefore, a useful term
for dealing with the titration of ampholytes and polyampholytes (e.g. proteins)
is the isoelectric point, pI. This is described as
the pH at which the effective net charge on a molecule is zero.
For the case of a simple ampholyte like the amino acid glycine the pI, when
calculated from the Henderson-Hasselbalch equation, is shown to be the average
of the pK for the a-COOH group and the pK for the a-NH2 group:
pI = [pKa-(COOH) +
pKa-(NH3+)]/2 Eqn.
20
For more complex molecules such as polyampholytes the pI is the average of
the pKa values that represent the boundaries of the zwitterionic form
of the molecule. The pI value, like that of pK, is very informative as to the
nature of different molecules. A molecule with a low pI would contain a
predominance of acidic groups, whereas a high pI indicates predominance of basic
groups.
back to
the top
Solvation and Hydration shells
Depending on the pH of a solution, macromolecules such as proteins which
contain many charged groups, will carry substantial net charge, either positive
or negative. Cells of the body and blood contain many polyelectrolytes (molecules that contain multiple same
charges, e.g. DNA and RNA) and polyampholytes that
are in close proximity. The close association allows these molecules to interact
through opposing charged groups. The presence, in cells and blood, of numerous
small charged ions (e.g. Na+, Cl-, Mg2+,
Mn2+, K+) leads to the interaction of many small ions with
the larger macroions. This interaction can result in a shielding of the electrostatic charges of like-charged
molecules. This electrostatic shielding allows macroions to become more closely
associated than predicted based upon their expected charge repulsion from one
another. The net effect of the presence of small ions is to maintain the
solubility of macromolecules at pH ranges near their pI. This interaction
between solute (e.g. proteins, DNA, RNA, etc.) and solvent (e.g. blood) is
termed solvation or hydration. The opposite effect to solvation occurs when the
salt (small ion) concentration increases to such a level as to interfere with
the solvation of proteins by H2O. This results from the
H2O forming hydration shells around the
small ions.
back to
the top
Role of the Kidneys in Acid-Base Balance
The kidneys function to filter the plasma that passes through the nephrons.
Filtration of the plasmas occurs in the glomerular capillaries of the nephron.
These capillaries allow the passage of water and low molecular weight solutes
(less than 70 kDa) into the capsular space. The filtrate then passes through the
proximal and distal convoluted tubules where reabsorption of water and many
solutes takes place. In the course of glomerular filtration and tubule
reabsorption the composition of the plasma changes generating the typical
composition of urine. From a biochemical standpoint the kidneys serve important
roles in the regulation of plasma acid-base balance and the elimination of
nitrogenous wastes.
back to
the top
Sodium Bicarbonate
Reabsorption
Regulation of plasma acid-base balance is primarily effected within the
kidney through control over HCO3- reabsorption and
secretion of H+. Secretion of H+, in excess of its
capacity to react with HCO3- in the tubular lumenal fluid,
requires the presence of other buffers (see below). The generation of
HCO3- and H+ occurs by dissociation of carbonic acid (H2CO3), formed in the
tubule cells from H2O and CO2, through the action of
carbonic anhydrase. Secretion of H+ into the lumen of
the tubule is accompanied by an exchange for Na+. This reabsorption
of Na+ occurs by an antiport mechanism during the exchange for H+.
Reduction in the intracellular concentration of Na+ occurs by an
active transport process involving a Na+/K+-ATPase pump
which pumps the excess Na+ into the interstitial fluid. The
intracellular HCO3- then diffuses from the tubule cell
into the interstitial fluid.
The capacity of the kidney to secrete H+ is regulated by the
maximal H+ gradient that can form between the tubule and lumen and
still allow transport mechanisms to operate. This gradient is determined by the
pH of the urine which in humans is near 4.5. The capacity to secrete
H+ would be rapidly reached if it were not for the presence of
buffers within the interstitial fluid. The H+ secreted into the
tubular lumen can undergo three different fates depending upon the concentration
of the three primary buffers of the interstitial fluid. These buffers are
HCO3-, HPO42- and NH3.
Reaction of H+ with HCO3- forms H2O
and CO2 which diffuse back into the tubule cell. The net result of
this process is the regeneration of HCO3- within the
tubule cell. This process is termed reabsorption of sodium bicarbonate. The
reabsorption of sodium bicarbonate takes place
primarily within the proximal convoluted tubules.
back to
the top
Excretion of Acid
As the concentration of HCO3- in the tubular lumen
drops the pH of the fluid drops due to an increasing concentration of
H+. The pH of the tubular fluid gradually approaches the
pKa for the dibasic/monobasic phosphate buffering system
(pKa = 6.8). The excess H+ reacts with dibasic phosphate
(HPO42-) forming monobasic phosphate
(H2PO4-). The
H2PO4- so formed is not reabsorbed and its
excretion results in the net excretion of H+. The greatest extent of
H2PO4- formation occurs within the distal
convoluted tubules and the collecting ducts.
back to
the top
Ammonia Secretion
Buffering of H+ is also accomplished by reaction with ammonia,
NH3, to form ammonium ion, NH4+. Elimination of
NH4+ is the major contributory factor in the ability of
the body to excrete acid. Because the pKa of
NH4+ is 9.3, excretion of acid in this form can be
accomplished without lowering the pH of the urine. Additionally important is the
fact that excretion of acid in the form of NH4+ occurs
without depleting Na+ nor K+.
Two principal reactions within tubule cells result in the generation of
NH3, conversion of glutamine to glutamate and conversion of glutamate
to a-ketoglutarate. These reactions are catalyzed by
glutaminase and glutamate dehydrogenase,
respectively (Equations 21 and 22).
Glutamine ------> Glutamate + NH4+ Eqn. 21
Glutamate -------> a-Ketoglutarate +
NH4+ Eqn. 22
Both of these enzymes are abundant in tubule cells. Ammonia is lipid soluble
and will diffuse down its concentration gradient out of the tubule cell into the
tubular fluid. There it reacts with H+ to yield
NH4+ which is excreted in the urine.
back to
the top
Neurotoxicity of Ammonia
Excess ammonia is severely neurotoxic. Marked brain damage is seen in cases
of failure to make urea via the urea
cycle or to eliminate urea through the kidneys. The result of either of
these events is a buildup of circulating levels of ammonium ion. Aside from its
effect on blood pH, ammonia readily traverses the brain blood barrier and in the
brain is converted to glutamate via glutamate dehydrogenase,
depleting the brain of a-ketoglutarate. As the a-ketoglutarate is depleted, oxaloacetate falls
correspondingly, and ultimately TCA cycle activity comes to a halt. In the
absence of aerobic oxidative phosphorylation and TCA cycle activity, irreparable
cell damage and neural cell death ensue.
back to
the top
Acidosis and Alkalosis
The kidneys play an important role in the control of acidosis by responding
with an increase in the excretion of H+. When H+ is
excreted as a titratable acid such as H2PO4- or
when the anions of strong acids such as acetoacetate are excreted there is a
requirement for simultaneous excretion of cations to maintain electrical
neutrality. The principal cation excreted is Na+. As the level of
excretable Na+ is depleted excretion of K+ increases. In
conditions of acidosis the kidney will increase the production of NH3
from tubular amino acids or amino acids absorbed from the plasma. As indicated
the NH3 can diffuse across the tubule cell membrane where it will
react with H+ to form the excretable ammonium ion without a
concomitant requirement for cation excretion. This demonstrates that an
inability of the kidney to generate NH3 would rapidly lead to fatal
acidosis.
When the kidneys fail to modulate HCO3- excretion,
metabolic alkalosis will develop. Alkalosis is normally countered quite
effectively by the kidney allowing HCO3- to freely escape.
Alkalosis generally only becomes problematic if the kidneys are restricted in
their ability to secrete HCO3-. This situation can occur
in patients taking diuretics since several of this class of drug cause a
reduction in the ability of the kidney to reabsorb an anion (e.g.
Cl-) concomitant with the reabsorption of Na+.
back to
the top
Back to Topics<<<<
This article has been modified by Dr. M. Javed Abbas.
If you have any comments please do not hesitate to sign my Guest Book.
20:35 21/12/2002
|
